Development of highly efficient carbon dioxide conversion system by applying the principles of biofuel synthesis
Expected to propose a new paradigm for electrochemical conversion of carbon dioxide

From left, Researchers Jun Ho Jang, Kyu Min Lee, and Professor Ki Tae Nam
Professor Ki Tae Nam's research team of the Department of Materials Science and Engineering developed the world's first electrochemical conversion of carbon dioxide that mimics the natural biofuel synthesis system and succeeded in synthesizing carbonate, a new concept of e-fuel (electricity-based fuel), from carbon dioxide.
The research team succeeded in establishing a new system for electrochemical conversion of carbon dioxide to synthesize biofuels. This system will introduce a new paradigm in the field of carbon dioxide conversion and utilization by developing a highly efficient system that can produce high value-added products with only a small cost in electrical energy.
Reducing the concentration of greenhouse gases in the atmosphere is currently a top priority for mankind. Countries around the world are tightening carbon regulations along with the 2050 Carbon Neutral Declaration. In order to implement carbon neutrality, it is not only essential to put in the effort to reduce carbon emissions, but also the technology to convert emitted carbon dioxide into high-value-added compounds.
As global responses to reduce greenhouse gas emissions is in its full swing, 'e-fuel', a carbon neutral fuel, has recently been in the limelight. ‘e-fuel’ is a technology that converts carbon dioxide into high value-added compounds by using electric energy. By utilizing eco-friendly energy such as solar and wind power as its energy source to reduce greenhouse gases and produce useful products, it is expected to be a core technology that can implement a carbon neutral society.
The technological principle of producing ‘e-fuel’ from existing carbon dioxide is as follows. As an example, when burning gasoline, gasoline molecules oxidize and turn into carbon dioxide, which powers the car's engine. Conversely, when carbon dioxide is reduced using electrical energy, electrons are injected into the carbon dioxide and converted into high energy fuels such as gasoline. Until now, through this reduction process alone, studies related to electrochemical conversion of carbon dioxide have produced materials such as carbon monoxide, formic acid and ethylene, which have higher energy than carbon dioxide. However, this method requires more electricity to be generated in order to obtain materials with higher energy. Therefore, the low economic feasibility of the commercialization of the electrochemical conversion of carbon dioxide leaves the industry and academia to be looking for a new breakthrough.
The research team presented a new breakthrough in 'e-fuel' production technology by applying the principle of synthesizing biofuels in the natural world to the electrochemical carbon dioxide conversion system. In the process of synthesizing biofuels, living organisms utilize the continuous electron transport flow created through the electron transport medium.
By mimicking the flow of such electrons, a new system for the electrochemical conversion of carbon dioxide has been developed. This system does not end with electrons being injected into carbon dioxide, but can escape again through the electron transport medium and flow continuously through the solution. As a result, a new methodology, completely different from those of conventional methods, was proposed to reduce carbon dioxide and made possible the synthesis of carbonate compounds from carbon dioxide. This is significant in that it has expanded the range of ‘e-fuel’ products that can be produced from carbon dioxide, which had previously been limited to products in reduced form.
The ‘e-fuel’ synthesis is an innovative system that allows the formation of high-value compounds with low electrical energy costs. Dimethyl carbonate (hereinafter DMC), synthesized by Professor Ki Tae Nam's research team, can be used as a fuel additive for gasoline and biodiesel, as well as various industries such as polymer manufacturing, medicine and batteries. Due to such high utility value, the market price of DMC is about three times higher than that of formic acid that can be obtained through the existing conversion of carbon dioxide.
On the other hand, the electrical energy required to produce DMC is 3.5 kWh/kg, which is similar to formic acid. Considering that the unit cost of solar power generation in 2030 is 94.2 won/kWh, the research team can produce 1 kg of DMC with an electric energy cost of about 330 won. It will soon be possible to take one step closer to the commercialization of electrochemical conversion of carbon dioxide through new development capable of synthesizing products with high market price compared to those of low electrical energy.
"This achievement is innovative in that it suggested a method for synthesizing carbonate compounds from carbon dioxide at room temperature using electric energy for the first time in the world, and is expected to play an important role in carbon dioxide reduction. By developing a new concept of a carbon dioxide conversion system that enables the synthesis of high value-added compounds with low electric energy cost, it is expected to present a new direction for electrochemical conversion of carbon dioxide in the future,” said Professor Ki Tae Nam.
The research was published online on July 9 in Nature Energy, a world-renowned international journal. Nature Energy is the world's most prestigious academic journals in energy, with the Impact Factor at 60.85 as of 2020.
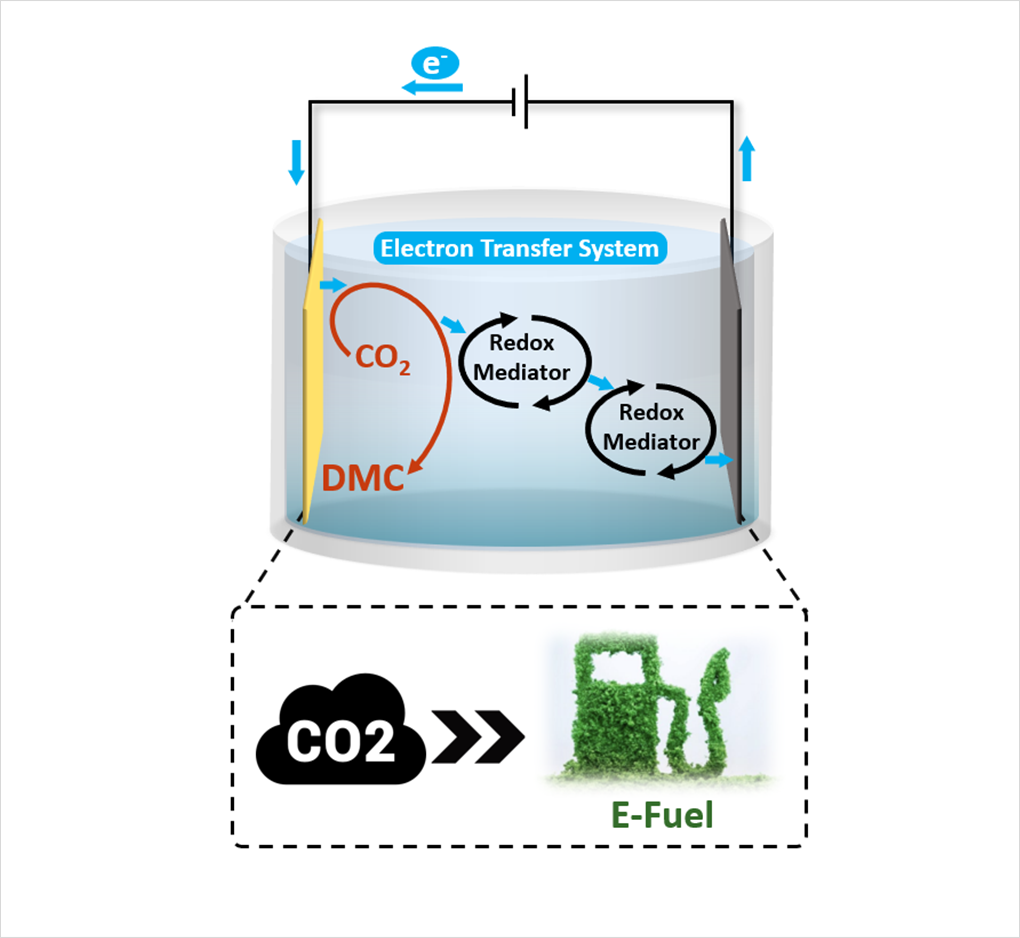
Figure Diagram showing the newly developed synthesis reaction of carbon neutral fuel(e-fuel)
For further information, please contact Prof. Ki Tae Nam.


